57. Functional quinoline-malononitriles for enhanced wound repair and monitoring
Wound healing is a complex biological process that, when disrupted, can result in chronic wounds and substantial healthcare burdens. Materials that enhance wound repair while enabling real-time monitoring represent a significant advancement in wound management. In this study, we report a series of fluorescent quinoline-malononitrile (QM) derivatives with aggregate-induced emission (AIE) properties and evaluate their therapeutic potential in dermal and epidermal cell models. Cytotoxicity assays revealed selective proliferative and anti-proliferative effects across different QM analogues. Notably, QOne (1) and MeQM (4) promoted keratinocyte proliferation, a critical step in re-epithelialisation. In a 3D in vitro wound model, both compounds significantly enhanced wound repair, accelerating the regeneration of the stratified epidermis and dermal matrix compared to untreated controls. Their intrinsic fluorescence also enabled real-time tracking of compound localisation during healing in a 3D in vitro wound model. These findings highlighted AIE-active molecules such as QOne (1) and MeQM (4) as promising dual-function agents for therapeutic intervention and fluorescence-guided wound monitoring.
56. Global genomic surveillance of tigecycline resistance gene tet(X) in Salmonella enterica
Abstract:
Objectives
The global transmission of plasmid-mediated tigecycline resistance gene tet(X) is a growing concern, yet its distribution in Salmonella remains poorly characterized. This study aimed to determine the prevalence and molecular features of tet(X) in Salmonella isolates from China and elucidate the global population structure and genomic surveillance framework.
Methods
A total of 865 Salmonella strains collected from diverse sources in China between 2019 and 2024 were screened for genetic analysis of tet(X), along with 89 tet(X)-positive Salmonella isolates from humans, animals, food, and the environment worldwide.
Results
One tet(X4)-positive S. 4,[5],12:i:- ST34 strain was recovered from pig feces among the 865 isolates. The tet(X4) gene was located on the commonly observed IncFIA8-IncHI1/ST17 plasmid. Globally, the 89 tet(X)-positive Salmonella isolates, collected between 2005 and 2025 originated from eight countries, predominantly the United Kingdom (44.94%) and China (22.47%). Most isolates were from humans (67.42%), followed by animals and foods. Among the four identified tet(X) variants, tet(X4) is the most prevalent, representing 83.15% of the isolates. All these isolates carried multiple antibiotic resistance genes and belonged to 16 serovars and 17 sequence types, primarily S. Kentucky ST198 (35.96%) and S. 4,[5],12:i:- ST34 (16.85%). Particularly, both S. Kentucky ST198 and S. 4,[5],12:i:- ST34 strains exhibited a close genetic relatedness within their respective lineages.
Discussion
The global distribution of tet(X)-carrying S. enterica strains across humans, animals, food, and the environment underscores a significant threat to public health. It highlights the urgent need for enhanced global surveillance of tet(X)-positive Salmonella within a One Health framework.
55. Water-based coupling of amino acids for sustainable solid-phase peptide synthesis
Solid-phase peptide synthesis (SPPS) is a primary source of peptides for structure–function studies in development of potential clinical, agricultural, veterinary or cosmetic applications. Despite its efficiency and scalability, SPPS produces unsustainable volumes of toxic waste and non-degradable solid supports. With regulatory changes now in effect to reduce solvent use, there is a pressing need for sustainable, water-based synthesis methods. Due to the aqueous insolubility of the commonly employed Nα-Fmoc-amino acids—the primary building blocks in SPPS—establishing water-based peptide synthesis protocols is extremely challenging. Here we show that amine salts of, for example, N-methylmorpholine or N,N,N-triethanolamine and Nα-Fmoc protected amino acids are water soluble at high concentrations. Activation with a pre-formed water-soluble activating agent enables high quality SPPS on a hydrophilic and biodegradable poly-ε-lysine-based solid support in aqueous solution without characteristic side reactions. The presented methodology provides a simple avenue to sustainable, scalable water-based chemical synthesis of peptides.
54. A Dual-Model Machine Learning Framework for Interpretable Design and Ensemble Prediction of C-Amidated Antimicrobial Peptides
Antimicrobial peptides (AMPs) offer promising alternatives to conventional antibiotics, yet most predictive models fail to account for chemical modifications that influence real-world efficacy. Among these, C-terminal amidation is a widely adopted and effective strategy that improves structural stability, membrane interaction, and protease resistance. In this study, we established an integrated framework for the design and prediction of C-terminal amidated AMPs targeting Escherichia coli. Our approach combined a design-oriented model based on an interpretable Explainable Boosting Machine (EBM), which extracts actionable sequence-level design rules, together with a reliable deployment model, built on a fine-tuned ESM2 deep learning architecture. The resulting tool, CAmidPred, enables both predictive classification and amino acid pattern analysis with outputs examined in relation to published alanine-scanning experiments. Using these models, we identified a pardaxin variant with improved activity against E. coli, demonstrating the practical utility of the dual-model framework in targeted AMP design.
53. Aggregation-Prone Antimicrobial Peptides Target Gram-negative Bacterial Nucleic Acids and Protein Synthesis
Aggregation of antimicrobial peptides (AMPs) enhances their efficacy by destabilising the bacterial cell wall, membrane, and cytosolic proteins. Developing aggregation-prone AMPs offers a promising strategy to combat antibiotic resistance, though predicting such AMPs and understanding bacterial responses remain challenging. Octopus bimaculoides, a cephalopod species, lacks known AMP gene families, yet its protein fragments were used to predict AMPs via artificial intelligence tools. Four peptides (Oct-P1, Oct-P2, Oct-P3, and Oct-P4) were identified based on their aggregation propensity. Among them, Oct-P2 reduced the viability of Escherichia coli and Staphylococcus aureus by up to 90%, confirmed by confocal laser scanning microscopy and scanning electron microscopy. It further aggregated plasmid DNA in vitro, and the presence of extracellular DNA reduced their antibacterial activity. With knockout mutants, it revealed that Oct-P2 was internalised into bacterial cells, possibly through membrane transport proteins, enhancing its antibacterial effect. Aggregation-induced emission assays and molecular dynamics simulations revealed that Oct-P2 aggregates with transcription promoter DNA, inhibiting transcription and translation in vitro. This dual-target mechanism not only highlights the potential of Oct-P2 as a lead template for new antimicrobial drug development, but also opens a new window for discovering AMPs from protein fragments against the upcoming challenge of bacterial infections.
52. Development of modern continuous flow solid phase peptide synthesis: chemistry, automation and sustainability impact
The invention of solid-phase peptide synthesis (SPPS) in the late 1950s marked the end of the era of laborious and technically difficult classical solution-phase assembly of these biomolecules. SPPS enabled the ready production of defined peptide sequences with unprecedented speed, reliability and scalability, revolutionising both fundamental biochemical research and therapeutic development. Over the following decades, iterative improvements in amino acid protecting group strategies, coupling reagents and instrumentation further cemented SPPS as a foundation of modern peptide science. This mini-review aims to provide a historical overview of the development and application of continuous-flow (CF) methods to SPPS, leading to its present status as the most effective means for the automated, sustainable, chemically efficient production of peptides. By integrating the principles of flow chemistry with solid-phase techniques, CF-SPPS delivers major gains in reagent economy, reaction kinetics and environmental impact. However, a major issue remains with the solid support itself. The continued reliance on resins originally optimised for batch processes and their corresponding unsuitability has restricted the full realisation of CF-SPPS’s potential. Thus, the development and implementation of next-generation polymer supports tailored specifically for CF processes represents a key frontier in the ongoing refinement of CF-SPPS.
51. Escherichia coli ST542 Co-Harbouring blaNDM-1 and mcr-1 in a Pig Farm, China
Carbapenems are potent antibiotics utilized to treat serious infections caused by multidrug-resistant (MDR) Gram-negative bacteria.1 However, the clinical use of carbapenems has led to the emergence and global dissemination of carbapenem-resistant Enterobacteriaceae (CRE), which pose a significant threat to effective clinical therapy.1 The genes encoding carbapenemases, particularly the New Delhi metallo-β-lactamase gene blaNDM, have been widely distributed among humans and detected in animals, food, and the environment.1,2 Colistin is considered as the last therapeutic resort for treating MDR Gram-negative bacteria, especially CRE bacteria.1,3 However, its efficacy has been undermined by the emergence of various antimicrobial resistance mechanisms, such as mutations in the two-component regulatory systems PmrA-PmrB and PhoP-PhoQ.3 Particularly, the global spread of plasmid-mediated colistin resistance gene mcr, first identified in 2015, has further exacerbated this issue.3 Here, we report the recent identification of Escherichia coli ST542 strains co-producing MCR-1 and NDM-1 isolated from a pig farm in Yangzhou, China.

50. Functionalized α-Cyanostilbene Derivatives for Detection of Hypoxia or Proteostasis Imbalance in Live Cells
α-Cyanostilbene represents one of the easily functionalized aggregation-induced emission (AIE) scaffolds. It has been widely adopted for the construction of fluorescent materials for broad applications. Here, we further expanded the utilization of α-cyanostilbene derivatives for the detection of hypoxia or proteostasis imbalance in live cells. Four different amine containing donors were introduced to construct α-cyanostilbene derivatives (R-ASC) with donor-acceptor scaffolds. Equipped with the cysteine (Cys) reactive group, maleimide (MI), R-ASC-MI shows fluorescence turn-on property upon binding with unfolded proteins in vitro and in live cells under proteostatic stress. By virtue of R-ASC-MI, the level of unfolded protein loads in cells can be quantified by flow cytometry, or visualized under microscope. Furthermore, we also characterized the performance of R-ASC-NO2, synthetic precursors of R-ASC-MI, in cellular hypoxia. R-ASC-NO2 revealed upregulated activities of nitroreductase, as well as increased hydrophobicity in live cells, under either chemical (NaN3) induced or atmospheric (1 % O2) hypoxia. Together, the advantages of easy modification and high signal-to-noise ratio of new α-cyanostilbene derivatives reported in this work highlight the great potential of α-cyanostilbene in constructing functional biosensors and many other domains.α-Cyanostilbene represents one of the easily functionalized aggregation-induced emission (AIE) scaffolds. It has been widely adopted for the construction of fluorescent materials for broad applications. Here, we further expanded the utilization of α-cyanostilbene derivatives for the detection of hypoxia or proteostasis imbalance in live cells. Four different amine containing donors were introduced to construct α-cyanostilbene derivatives (R-ASC) with donor-acceptor scaffolds. Equipped with the cysteine (Cys) reactive group, maleimide (MI), R-ASC-MI shows fluorescence turn-on property upon binding with unfolded proteins in vitro and in live cells under proteostatic stress. By virtue of R-ASC-MI, the level of unfolded protein loads in cells can be quantified by flow cytometry, or visualized under microscope. Furthermore, we also characterized the performance of R-ASC-NO2, synthetic precursors of R-ASC-MI, in cellular hypoxia. R-ASC-NO2 revealed upregulated activities of nitroreductase, as well as increased hydrophobicity in live cells, under either chemical (NaN3) induced or atmospheric (1 % O2) hypoxia. Together, the advantages of easy modification and high signal-to-noise ratio of new α-cyanostilbene derivatives reported in this work highlight the great potential of α-cyanostilbene in constructing functional biosensors and many other domains.
49. Dimerization and lysine substitution of melittin have differing effects on bacteria
Introduction: Melittin is a potent antimicrobial peptide from bee venom that is effective against both Gram-positive and Gram-negative bacteria. However, it is extremely toxic to mammalian cells and, as yet, has no clinical use. Modifications to its amino acid sequence, cyclization, truncation, and dimerization have been attempted in order to reduce its toxicity whilst maintaining its antimicrobial activity.
Methods: In this study, we targeted the three lysine residues present in melittin and substituted them with lysine homologs containing shorter side chains (ornithine, Orn, diaminobutyric acid, Dab, and diaminopropanoic acid, Dap) and made both parallel and antiparallel melittin dimers to observe how lysine substitution and dimerization affects its activity and toxicity. The antibacterial activity of melittin and its analogs was tested against S. aureus (Gram-positive bacteria) and E. coli (Gram-negative bacteria), and cytotoxicity was tested against the mammalian cell lines HEK293 and H4IIE.
Results: Overall, dimerization and lysine substitution exhibited improved antimicrobial activity toward E. coli and limited improvement toward S. aureus. However, mammalian cell toxicity was only marginally reduced compared to native melittin. Interestingly, the parallel dimer was found to be marginally more active than the antiparallel dimer, indicating orientation maybe important for activity, although both dimers were less effective than the native and Lys-analog peptides toward S. aureus. Of the Lys substitutions, Dab and Dap improved melittin’s activity toward E. coli.
Discussion: Dimerization and Lys substitution of melittin improved the antimicrobial activity toward Gram-negative bacteria but did not significantly improve its activity toward Gram-positive bacteria. Some analogs also displayed reduced toxicity toward HEK293 and H4IIE cells but overall remained toxic at bactericidal concentrations. Our data indicates that although highly antibacterial, melittin’s toxicity is the major drawback in its potential use.
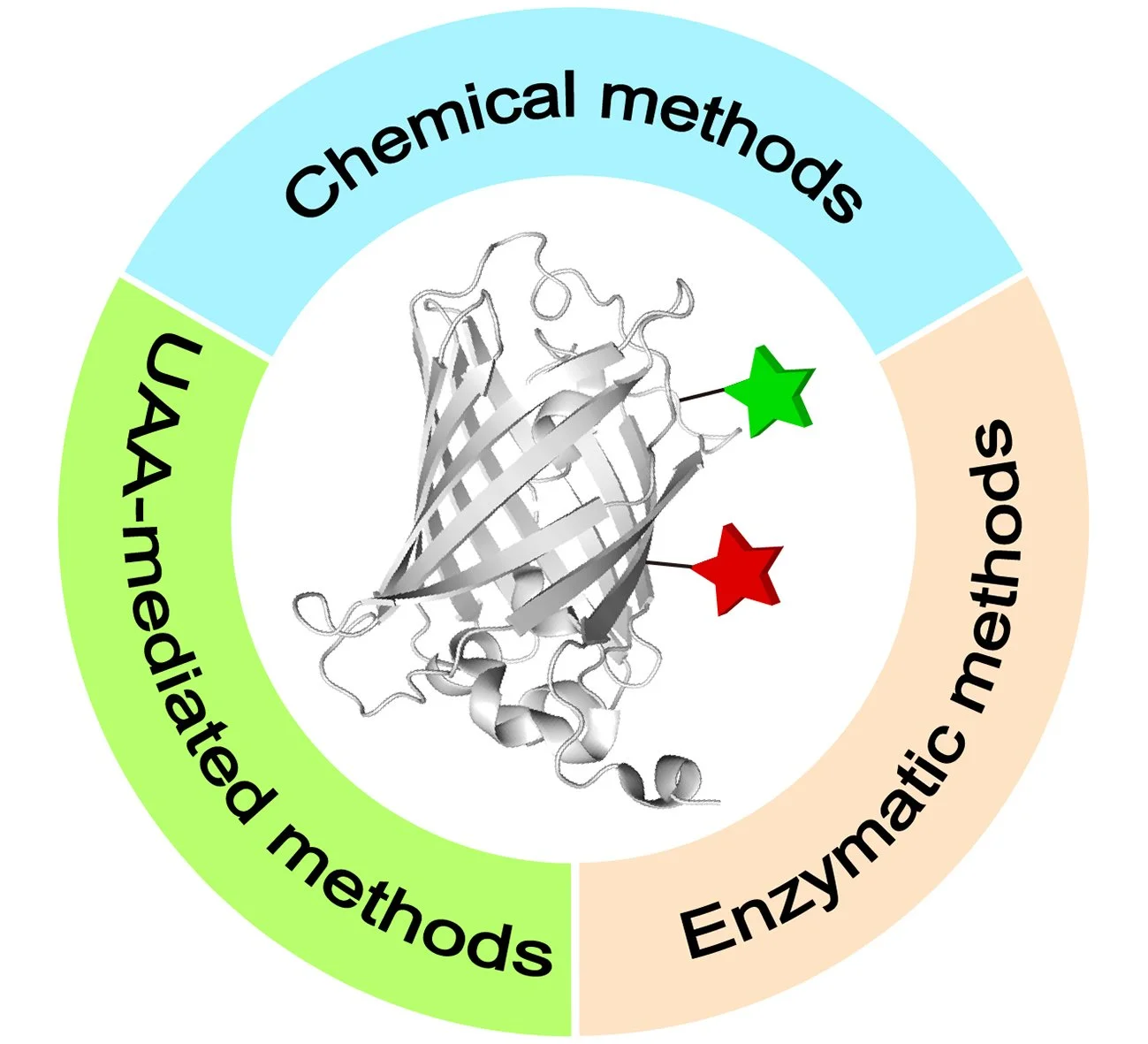
48. Recent Toolboxes for Chemoselective Dual Modifications of Proteins
Site-selective chemical modifications of proteins have emerged as a potent technology in chemical biology, materials science, and medicine, facilitating precise manipulation of proteins with tailored functionalities for basic biology research and developing innovative therapeutics. Compared to traditional recombinant expression methods, one of the prominent advantages of chemical protein modification lies in its capacity to decorate proteins with a wide range of functional moieties, including non-genetically encoded ones, enabling the generation of novel protein conjugates with enhanced or previously unexplored properties. Among these, approaches for dual or multiple protein modifications are increasingly garnering attention, as it has been found that single modifications of proteins are inadequate to meet current demands. Therefore, in light of the rapid developments in this field, this review provides a timely and comprehensive overview of the latest advancements in chemical and biological approaches for protein dual functionalization. It further discusses their advantages, limitations, and potential future directions in this relatively nascent area.