55. Water-based coupling of amino acids for sustainable solid-phase peptide synthesis
Abstract:
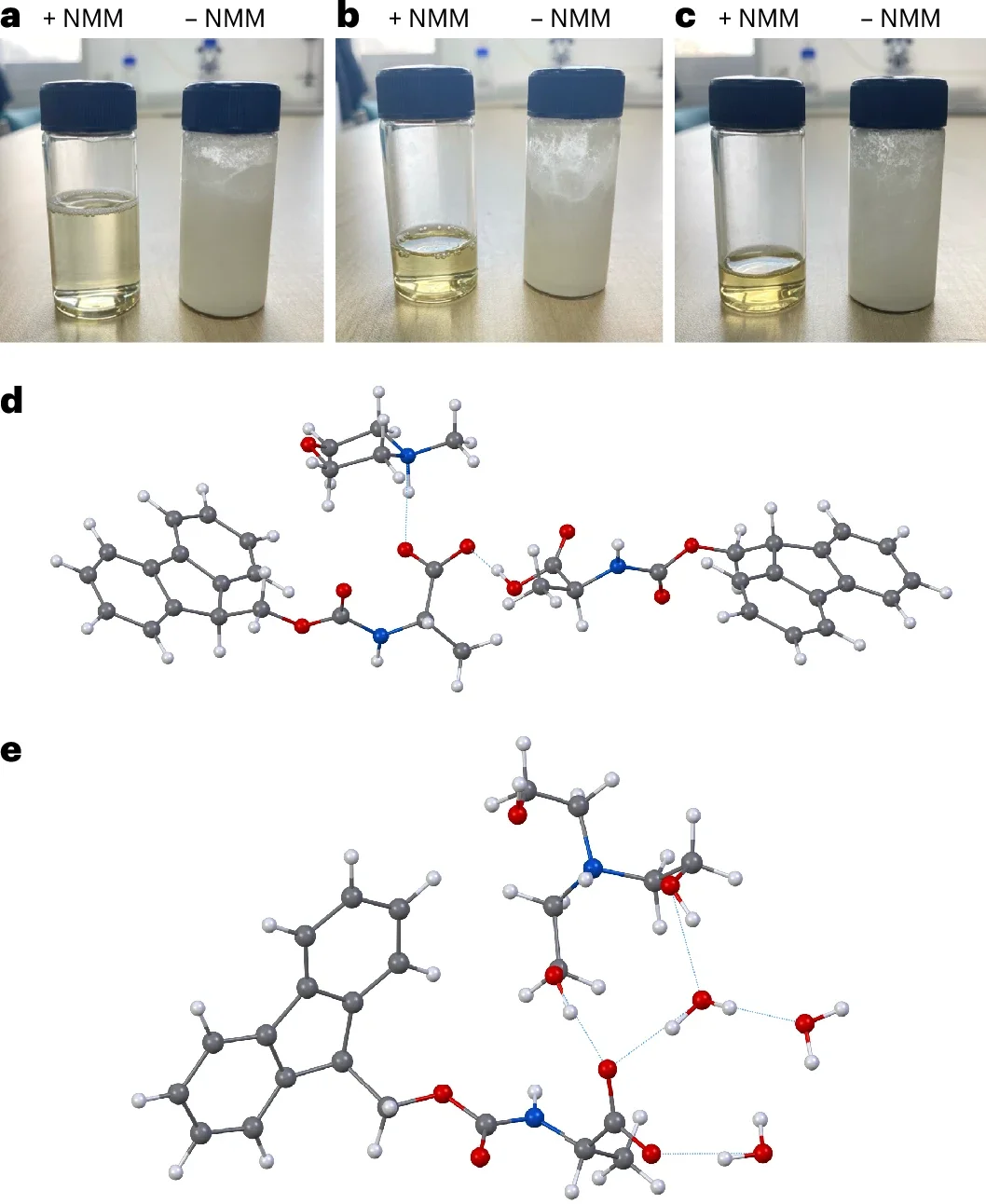
Solid-phase peptide synthesis (SPPS) is a primary source of peptides for structure–function studies in development of potential clinical, agricultural, veterinary or cosmetic applications. Despite its efficiency and scalability, SPPS produces unsustainable volumes of toxic waste and non-degradable solid supports. With regulatory changes now in effect to reduce solvent use, there is a pressing need for sustainable, water-based synthesis methods. Due to the aqueous insolubility of the commonly employed Nα-Fmoc-amino acids—the primary building blocks in SPPS—establishing water-based peptide synthesis protocols is extremely challenging. Here we show that amine salts of, for example, N-methylmorpholine or N,N,N-triethanolamine and Nα-Fmoc protected amino acids are water soluble at high concentrations. Activation with a pre-formed water-soluble activating agent enables high quality SPPS on a hydrophilic and biodegradable poly-ε-lysine-based solid support in aqueous solution without characteristic side reactions. The presented methodology provides a simple avenue to sustainable, scalable water-based chemical synthesis of peptides.
Wellings, D.; Greenwood, J.; Thomas, I.; Hughes, C.; Li, W.; Lin, F.; Hossain, M.A.; Lanza, A.; Meldal, M.; Wade J.D. (2026) Water-based coupling of amino acids for sustainable solid-phase peptide synthesis, Nature Sustainability, DOI: 10.1038/s41893-025-01761-z